Contents | 1. Introduction to structural design | 2. Loads | 3. Wood | 4. Steel |
Introduction to reinforced concrete |
Concrete consists of aggregate (course aggregate, or gravel, and fine aggregate, or sand) that is bound together with hydraulic cement. "Hydraulic" refers to the fact that the cement reacts with and hardens (cures) in the presence of water. Water is therefore the third necessary ingredient in plain concrete.
While the Romans used all sorts of aggregate in their 2000-year-old plain concrete structures — including recycled brick rubble, tile, and relatively large hunks of rock — modern aggregate consists almost exclusively of stone and sand that are combined in precise proportions, using various sizes or grades to minimize voids between the aggregate particles. The logic of grading can be understood by imagining a container filled with spherical stones, all of the same size (Figure 5.1a). Clearly the voids left between the spheres can be filled using smaller stones (Figure 5.1b); and the voids between those smaller stones can be filled with even smaller stones, or sands. The goal is to minimize such voids for two primary reasons. First, cement — which fills the voids and binds the aggregate together — is far more expensive than gravel and sand. Second, cement tends to shrink as it cures, so that large quantities of cement would result in unacceptably large amounts of shrinkage and cracking. Using only large stones is clearly less effective than adding a variety of smaller aggregate to a mix that includes large stones. On the other hand, using only small aggregate is also less effective, since a larger stone could displace not only a great deal of the smaller aggregate but also eliminate the voids (cement) between the small aggregate pieces that have been displaced.

So why not use really large rocks as aggregate, thereby displacing the most voids? In fact, the largest aggregate size used in any given mix is limited both by the spacing between bars of reinforcing steel (since the aggregate must be able to easily pass through the matrix of steel bars) and by the smallest dimension of the formwork or slab thickness. For this reason, maximum aggregate sizes of 3/4 in. or 1 in. are fairly common. Aggregates graded in this way typically comprise about 60 to 80 percent of the volume (and 70 to 85 percent of the weight) of plain concrete, and consist of several grades of fine aggregate (sand) and several grades of course aggregate (gravel), as represented in Figure 5.1c.
Modern cements — at least since 1824 when Joseph Aspdin patented a hydraulic mix whose color was similar to that of a stone found on the Isle of Portland in the English Channel (hence, portland cement) — contain a number of ingredients (calcium, silicon, aluminum and iron) which are ground, blended, and heated in a kiln to create a cement clicker which, in turn, is blended with gypsum and ground again into a fine powder. Cement ingredients come from rather commonplace materials such as limestone, shells, shale, clay, sand and iron ore.
Blended cements combine portland cement with other hydraulic materials including industrial by-products such as fly ash, blast furnace slag, and silica fume. These products can actually increase the durability and strength of concrete by reducing the amount of water necessary for the workability of the concrete mix. It turns out that more water is used in concrete than is needed for the chemical reaction between water and cement that results in the hardening of the cement; this "extra" water is required so that the concrete can be mixed and placed into forms; otherwise, it would be too stiff. When this extra water eventually evaporates, it leaves pores in the concrete that reduce the strength of the concrete (since a void cannot resist tensile or compressive forces) while providing a pathway for salts or other unwanted elements into the concrete, leading to various forms of damage or distress. Certain materials, such as fly ash — a by-product of coal-fired electric generating plants — are unusually smooth and spherical. For that reason, they act like ball bearings within the concrete mix, facilitating mixing and placing with reduced quantities of water.
The strength of concrete is quite sensitive to the water-cement (w/c) ratio: too little water makes the concrete hard to place, and encourages honeycombing — i.e., the appearance of voids when the formwork is stripped because the concrete was too stiff to fill the form properly, thereby reducing its strength. On the other hand, too much water leads to loss of strength and durability as described earlier. For ordinary concrete, a w/c ratio of about 0.6 (comparing the weights of the two materials) is typical, although in corrosive environments, the w/c ratio should be reduced to 0.5 or even 0.4. By using blended cements, the w/c ratio can be lowered — reducing voids and increasing both strength and durability — without compromising the workability of the mix. One also scores points with green building rating systems, since the use of recycled materials such as fly ash is considered beneficial. Implicitly acknowledging the impact of concrete on the production of global warming gases, the ACI, beginning in 2014 and continuing in 2019, has included a short, and rather passive-aggressive, section on sustainability in their Building Code Requirements for Structural Concrete and Commentary. After stating that "the licensed design professional shall be permitted to specify in the construction documents sustainability requirements in addition to strength, serviceability, and durability requirements of this Code," the next section gratuitously reminds us that "strength, serviceability, and durability requirements of this Code shall take precedence over sustainability considerations."
There are some downsides to the use of such recycled material to replace a portion of the portland cement. In general, concrete cures faster when only portland cement is used, as blended cements slow down the curing process: this slower cure time could result in substantial losses (since time = money), especially in multistory buildings where each floor is cast sequentially only after the prior floor structure has cured sufficiently. Additionally, some of these recycled ingredients, such as silica fume, have actually become more expensive than standard cement; and some of the lower-priced options may contain unwanted ingredients (e.g., sulfur) that could increase the risk of cracking or corrosion of steel reinforcement.
Table 5.1:Types of portland cement
| Name | Common applications | |
|---|---|---|
| Type I | General purpose | For ordinary construction of buildings, pavement, etc. |
| Type II | Moderate sulfate resistance | For concrete exposed to sulfate ions in soil or water |
| Type III | High early strength | For use in cold weather, or where rapid construction is desired |
| Type IV | Low heat of hydration | Rarely used; formerly used in massive structures like dams |
| Type V | High sulfate resistance | For concrete exposed to high levels of sulfate ions in soil or water |
Five types of portland cement are manufactured, as shown in Table 5.1. In practice, Types I and II are often combined into a single product (Type I/II). Type III is more finely ground, which tends to accelerate the mostly exothermic chemical reactions associated with curing, providing more heat to keep these reactions going in cold weather. Type IV is no longer commonly used, but was designed to slow down the exothermic reactions associated with curing in order to minimize temperature differentials in massive structures like dams (Type II cement is now more commonly used for such purposes, as its moderate heat of hydration has proven to be acceptable in massive concrete construction). Type V is like Type II, but with even more resistance to sulfate attack.
Water used in reinforced concrete should be free of organic matter and salts. Ordinary municipal water works well and is commonly used, but some forms of recycled water are also acceptable. When concrete is cast in either extremely hot or extremely cold conditions, ice may be added to the water, or the water may be heated, to compensate for these environmental conditions. This adds some expense to the process, but otherwise helps maintain an appropriate rate of curing.
Aside from the three main constituents of concrete — aggregate, cement, and water — other ingredients, called admixtures, are sometimes added to the concrete mix. The most commonly used are air-entraining agents. These admixtures increase the amount of air in concrete by forming tiny bubbles that are spread throughout the mix, allowing freezing water to expand (into these bubbles) without cracking the concrete. For this reason, air entrainment is almost always specified when concrete may be subject to freezing conditions. In fact, most cement types are available with such air-entraining agents already included.
Water reducers constitute a second class of admixtures that includes both mid-range and high-range variants, the latter also known as plasticizers or superplasticizers. These admixtures create workable, and in some cases highly "flowable," concrete at lower w/c ratios, thereby increasing the concrete's strength and durability. Some of these admixtures, such as fly ash, are precisely the ingredients used to create blended cements.
Corrosion inhibitors are a third type of admixture. These are designed to protect reinforcing steel from rusting in particularly corrosive environments; otherwise the inherently alkaline nature of concrete protects steel bars from corrosion without requiring any added ingredients.
Lack of ductility is an undesirable property for a structural material, and is associated with low tensile strength. This is because small cracks or imperfections in a brittle material tend to propagate when pulled apart under tension, leading to a characteristic type of brittle failure. To prevent sudden, catastrophic failure of reinforced concrete beams, for example, the amount of reinforcement must be kept small enough so that the steel will yield (in a ductile manner) before the concrete crushes (in a sudden, catastrophic, brittle manner). Where this is not possible — in structural elements controlled by compression — safety factors must be adjusted accordingly.
Concrete mixes are designed to achieve a desired compressive strength with the minimum amount of cement compatible with adequate workability. The primary variable in determining strength is the ratio of water to cement (w/c), where this ratio is based on the relative weight, not volume, of the two materials. A methodology for proportioning the constituent ingredients in concrete promulgated by the American Concrete Institute (ACI) is widely used in practice.
Concrete, however, poses unique challenges as a material because the constituent ingredients are often stored and put together at a batching plant, and then delivered to the job site in special trucks containing rotating drums in which the ingredients are mixed in transit. When the concrete is then cast (not "poured") into forms, there is no way to validate the strength of the mix unless portions of the mix are set aside and tested. Some tests can be performed immediately on the job site: for example, the percentage of entrained air in the mix can be measured with an air entrainment meter (Figure 5.2a) and the consistency of the mix — and by implication the workability — can be evaluated with a slump test (Figure 5.2b). The first of these tests is based on Boyle's law, with air pressure and volume being inversely proportional. The second test relies less on scientific equations and more on low-tech empirical evidence — when fresh concrete is placed in an inverted 12-in.-high cone and the cone is quickly removed, the concrete will "slump" down to an extent that can be correlated with its consistency and workability (which is, to some extent, related to the w/c ratio). A more accurate w/c measurement can be obtained by conducting a microwave oven test that determines this ratio by essentially heating a cylinder of fresh concrete until the water evaporates and then comparing the weight of evaporated water to that of the cement.

A test that is specified by the American Concrete Institute (ACI) and often mandated by building codes is a compressive strength cylinder test. At least two 6 × 12 cylinders — or three 4 × 8 cylinders — are filled with fresh concrete at the job site and cured in a testing lab for 28 days (Figure 5.2c). At that time, they are crushed in a testing machine and their compressive strength is measured. Two cylinders are filled for every 150 cubic yards of concrete (or for every 5,000 ft2 of slab surface) and, after 28 days of controlled curing, the average strength of any three consecutively tested cylinders must be greater or equal to the specified cylinder strength of the concrete. Alternatively, the concrete is considered acceptable — even if this first criterion is not met — as long as all tested cylinders are no more than 500 psi weaker than the specified cylinder strength.
Aside from concrete's relatively high compressive strength, the material properties that are most significant in terms of its structural behavior are low tensile strength, brittleness, and shrinkage.
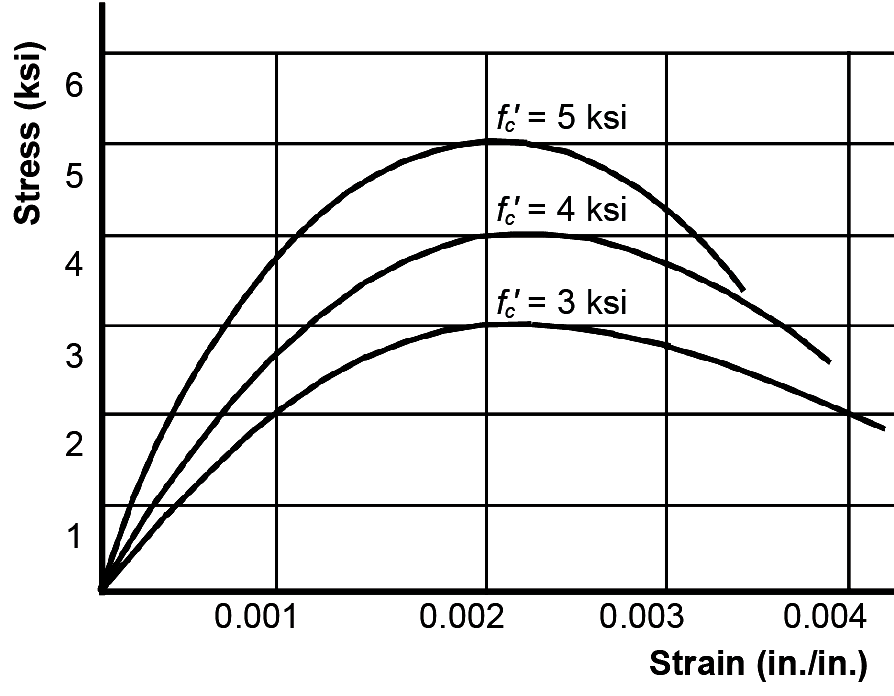
The specified compressive strength of concrete, fc', is generally in the range of 3000 – 5000 psi for ordinary projects (Figure 5.3), although much higher values can be obtained, at greater expense, especially for high-rise applications. This compressive strength is not achieved immediately after the concrete is mixed, but only after 28 days of curing. Of course, if 28 days of waiting were required before a story could be cast on top of the story immediately below, completing the structure for a 12-story cast-in-place reinforced concrete building would take more than a year. In fact, concrete typically reaches more than half its specified strength after a week; at this time, formwork can be stripped and the next floor can be cast, even though the specified concrete strength has not yet been achieved. Concrete continues to get stronger for several years after the initial 28-day curing period, but this added strength is not considered explicitly in the design process.
Low tensile strength makes plain concrete unsuitable for most structural applications, since even elements subjected to compressive stresses generally need sufficient tensile strength to inhibit buckling. For this reason, and to extend the range of its applications to beams and slabs as well as columns, concrete is reinforced with steel reinforcing bars, or "rebars," in regions of the cross section where tension is expected to occur.
Concrete — actually cement within the concrete mix — shrinks as part of the curing process. Expansion and contraction due to temperature changes can also cause differential movement between concrete and adjacent materials. To reduce and control cracking in slabs where this movement is restrained, for example by perimeter beams containing steel reinforcement, additional temperature and shrinkage reinforcement is added to 1-way slabs. These are slabs where the concrete spans and is structurally reinforced in one direction only, so that temperature and shrinkage reinforcement must be placed perpendicular to this spanning direction. In beams, columns, or two-way slabs, no temperature and shrinkage reinforcement is needed since reinforcement is already present where cracking might otherwise occur.
Creep is a form of inelastic shrinkage of concrete. As opposed to elastic deformations, which are proportional to loads and reversible (i.e., the deformation goes away when the load is removed), creep is neither proportional to the load, nor is it entirely reversible. Instead, it represents an additional, and ongoing, shrinkage of concrete under load above and beyond the chemically-induced curing shrinkage mentioned earlier, or the elastic deformation under load described in Equation 1.18. For most beams and slabs, this phenomenon can be safely ignored; for multi-story buildings, however, the effects of creep can become important, especially when adjacent lower-story columns or shear walls experience different amounts of creep owing to different loading or reinforcement conditions. Such analysis is, however, beyond the scope of this book.
In an ideal environment, the alkaline nature of concrete protects the steel reinforcing bars within from corrosion. In real environments, several problems may affect the durability of reinforced concrete structures.
Chloride-induced corrosion of reinforcing bars may occur for various reasons. The most obvious causal agents are de-icing salts that could get onto the surface of concrete slabs, e.g., in parking garages, and work their way into the concrete through cracks or pores. Remarkably, some older concrete has experienced this type of corrosion because of agents deliberately added to the concrete — either as "accelerating admixtures" to speed up the curing process or "etching agents" to remove cement at the surface of the concrete in order to reveal a more textured aggregate pattern.
Carbonation is a chemical process that occurs when cement reacts with "acid rain," causing the concrete to lose some of its alkalinity — this reduces the protection that the concrete ordinarily provides to reinforcing steel, and could lead to corrosion where concrete surfaces are exposed to such environmental conditions.
Sulfate attack is to some extent mitigated by the use of appropriate cements (Types II or V). Otherwise, where concrete comes into contact with sulfates in ground water or from ingredients in the concrete mix — e.g., some types of blast furnace slab or pulverized fuel ash — not only is alkalinity reduced (as in carbonation), but chemical reactions involving sulfates and concrete ingredients cause a volume increase that can lead to spalling of the concrete.
Alkali-silica reaction (ASR) occurs when alkalis in portland cement (or from other sources) react with certain aggregate in the concrete mix, forming an alkali-silica gel that expands — leading to cracking.
Precast concrete is reinforced concrete that is cast away from the building site, and assembled on site. Some (but not all) precast concrete is available in standard shapes and dimensions: floor and roof planks, tees and double-tees are examples. Otherwise, precast concrete may be fabricated in any shape and size consistent with the laws of statics, the strength and stiffness of the materials, and the constraints imposed by formwork, transportation, handling, and erection.
Precasting may imply a loss of structural continuity if connections are made with steel inserts bolted or welded together to create simple supports. On the other hand, it is possible, through emulative detailing, to design precast systems whose behavior is identical to that of site-cast systems. This is done by maintaining the continuity of steel reinforcement from element to element. Special products are available to connect rebars that have been left exposed at the ends of the concrete pieces; non-shrinking grouts are then used to fill in the voids and complete the structural connection.
Precast concrete is also widely used as "nonstructural" cladding in addition to being used as primary structure, especially in the US. The quotation marks around nonstructural hint at the inadequacy of the term: in fact, all cladding is structural since it must resist wind, seismic, and impact loads and transfer these loads to the primary lateral-force resisting structural system of the building.
Autoclaved aerated concrete (AAC) has some of the ingredients characteristic of plain concrete — cement, fine aggregate, and water — but also contains aluminum powder that reacts chemically to release hydrogen gas. This gas creates a kind of foam out of the concrete mix, which is easily cut into blocks or other shapes and then hardened within a pressurized autoclave chamber. The resulting product is lightweight, the entrapped air makes it highly insulative, and the units can be stacked and mortared much like more conventional concrete unit masonry.
Shotcrete is a modified mixture of concrete or mortar that is pneumatically "shot" from a hose at high velocity directly onto a surface, requiring no formwork (other than the surface onto which it adheres) or consolidation. There are two primary variants: a wet-mix where the dry ingredients are already mixed with water, and a dry-mix, where water is combined with the dry ingredients at the nozzle (sometimes called gunite).
© 2020 Jonathan Ochshorn; all rights reserved. This section first posted November 15, 2020; last updated November 15, 2020.